By gaining three electrons, nitrogen (N) turns into an ion with an ionic charge of -3 (N 3-). In order to fill its outer shell, nitrogen needs to gain three electrons. It has 7 electrons in total, with 5 of them on the outermost shell. 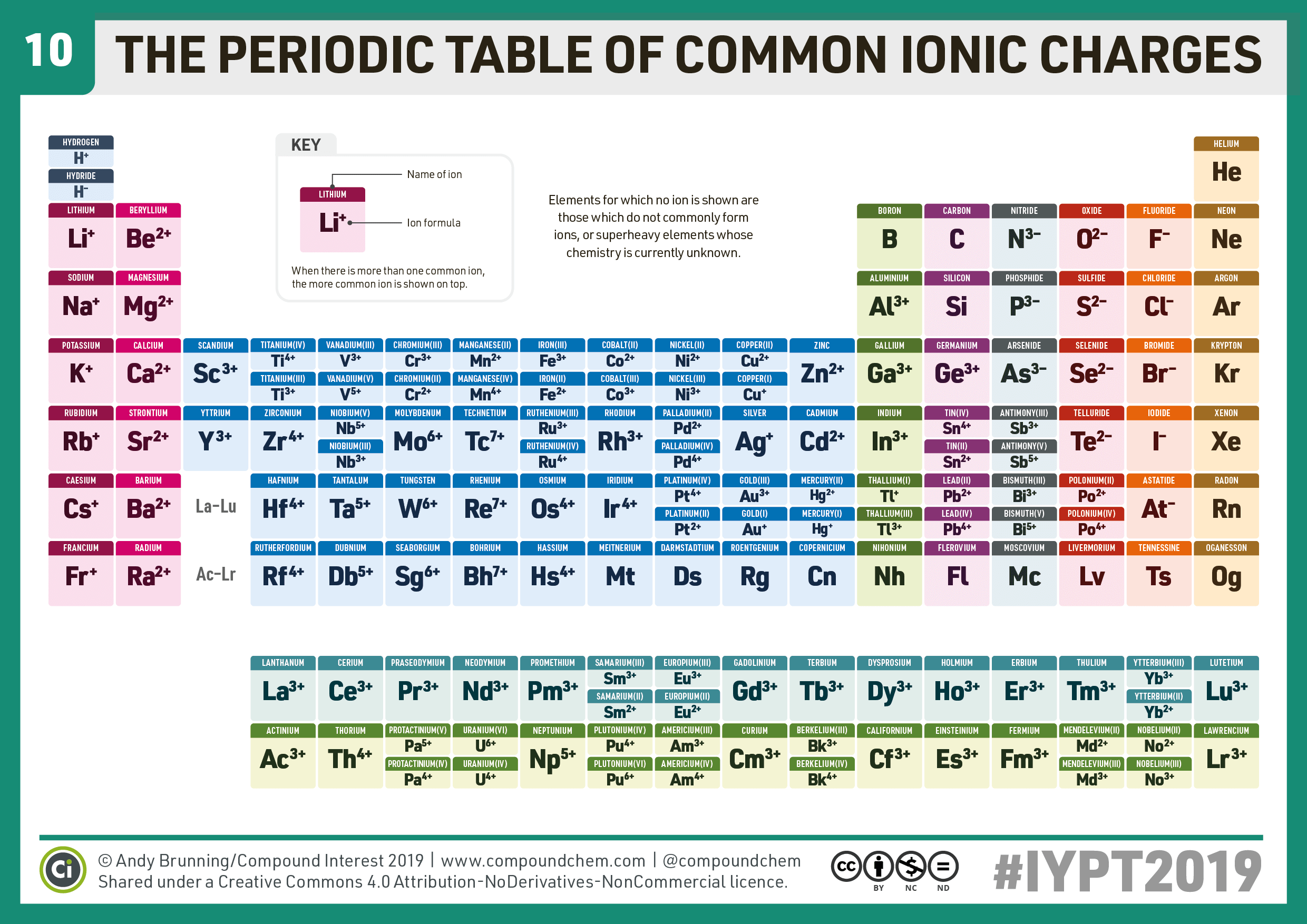
Nitrogen is a nonmetal found in group 15. Question 2: How many electrons does nitrogen (N) need to gain/lose to form an ion? So, strontium (Sr) will lose these two electrons to form a strontium ion with a +2 charge (Sr 2+)! There are two electrons in the outermost shell of all group 2 elements, and they want to get rid of them in order to achieve a more stable configuration. Notice that strontium is a part of group 2. Ions which ended with -ide could be either monatomic or polyatomic. Note that any time in ion hasn’t ending of -ate or -ite it is a polyatomic ion. The first step is to look at the periodic table to see in which group strontium (Sr) is found. Nitride is a monatomic ion, so you would consult the periodic table to determine its formula and charge. Question 1 : How many electrons does strontium (Sr) need to gain/lose to form an ion? To finish off, let's look at some questions involving ionic charge. Electrons equal proton number in neutral atoms. Remember: An element's atomic number is its number of protons. On the other hand, when an atom gains extra electrons to form an anion, its resulting ionic mass is higher. When an atom loses electrons to form a cation, its resulting ionic mass is lower. This difference in atomic and ionic size is mainly to the electrons being added or removed to form the ions. In the periodic table, the general trend in ionic radius is as follows: positively charged ions (cations) tend to have a smaller radius compared to their parent atoms, whereas negatively charged ions (anions) tend to be bigger than their parent atoms. 
For example, when Lithium loses an electron to form Li + ion, its radius changes from 145 pm to 76pm. When a neutral atom gains or loses electrons, its radius changes in size. For example, sodium ion (Na +) helps regulate and control body fluids, potassium (K +) ions helps regulate body fluids and cellular functions, Calcium ions (Ca 2+) is important in muscle contraction, whereas magnesium ions (Mg 2+) is essential for certain enzymes, muscles, and nerve control! Periodic Trends: Ionic Radius Did you know that there are many ions that are very important to our bodies? These ions play a vital role in different physiological and metabolic functions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
